Postdoctoral Work: Environmental Chemical Kinetics
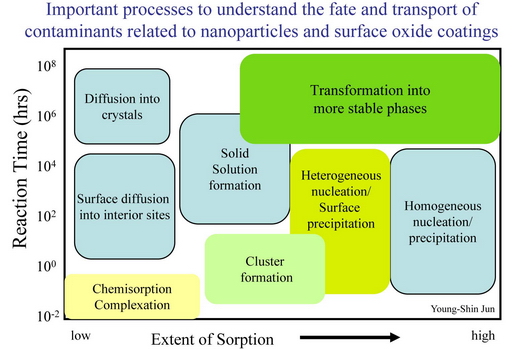
Environmental nanoparticles are often poorly-crystalline or metastable structures, whose kinetics of formation and growth are poorly understood. Further, the sorption or growth of nanoparticles on mineral surfaces may control the mineral surface’s reactivity and modify its ability to influence the fate and transport of contaminants. Due to the characteristic length scale, a holistic understanding of the nucleation mechanisms and kinetics of nanoparticle formation on mineral surfaces is difficult to achieve with traditional methodology. In this work, my aim is to determine the molecular nature and mechanisms of nucleation on surfaces, the kinetics of nanoparticle nucleation and growth, and the effect of surface topology and the presence of heavy metal ions or organic compounds on these processes using newly developed synchrotron-based techniques along with the conventional surface sensitive techniques. These results will help us understand the connection between molecular-scale studies and field-scale experimental data and provide more accurate predictive models of the fate and transport of environmental contaminants.
Research Topics
- Nucleation, Growth, and Transformation of Iron Oxide Nanoparticles on Mineral Surfaces
1. Developed the new grazing incidence small angle X-ray scattering (GISAXS) setup for the environmental applications (under dry conditions and in aqueous systems).
2. Conducted the first environmental applications of in situ time resolved GISAXS.
3. Collected the kinetic data of nucleation and growth of iron oxide nanoparticles (from a few nanometer sizes) in aqueous systems. This is the first kinetic data to resolve the nucleation and growth of nanoparticles in situ in aqueous systems (with better statistics than any techniques and shorter data acquisition time).
Young-Shin Jun, Glenn A. Waychunas, Michael F. Toney, Byeongdu Lee, and John Pople, “Characteristics of Environmental Iron Oxide Nanoparticles on the (10-10) Quartz Surfaces: First Environmental Applications of Grazing Incidence Small Angle X-ray Scattering,” In Preparation.
- Anion Adsorption on Iron Oxide Surfaces
1. Analyzed the silicate sorbed hematite crystal truncation rods data and compared the silicate sorption geometry on hematite with that of arsenate.
2. Collected the first Si K-edge grazing incidence extended x-ray absorption fine structure (GIEXAFS) spectra.
3. Analyzed the silicate adsorption geometry at the surface by combining CTR and GIEXAFS data. The complexed silicate on the hematite (1-102) surface is linked by a single oxygen to surface Fe, i.e. a monodentate connection, with an interatomic Si-Fe distance close to those observed in the nontronite and acmite structures (which are natural Si-Fe containing minerals). This is the first evidence that identifies silicate as a well-defined sorption complex rather than only as an amorphous surface precipitate. These findings have important significance for the nature of passivation processes on Fe oxides and other important reactive natural surfaces.
Glenn A. Waychunas, Young-Shin Jun, Peter J. Eng, Sanjit K. Ghose, and Thomas P. Trainor, “Anion Sorption Topology on Hematite Surfaces: Comparison of Arsenate and Silicate.” Adsorption of Metals to Geomedia II (Peer-Reviewed Book Chapter), Barnett, M.O and D.B. Kent, Editors, Elsevier, 2008.
- Effect of Exopolymeric Saccharides on Nanoparticle Growth at the Mineral-Water Interfaces
Recent Poster
Environmental Remediation Science Program PI Meeting
April 2007Molecular-level Investigations of Nucleation Mechanisms and Kinetics of Formation of Environmental Nanoparticles
(click here to see the poster)
Experimental Approaches
- Aquatic Chemistry: Aqueous conditions (Ion species and Complexation)
- Microscopic Surface Observations
Atomic Force Microscope (AFM) Transmission Electron Microscope (TEM)
- Synchrotron-Based Measurements: Investigation about Interfacial Reactions
Grazing Incidence Small Angle X-ray Scattering
(SSRL BL1-4 and BL 4-2 and APS 12ID)Small Angle X-ray Scattering
(SSRL 1-4)Grazing Incidence EXAFS and XANES
(ALS BL 6.3.1, APS sector 4 ID and 13ID, NSLS BL X15B, and SSRL BL11-2)Crystal Truncation Rod
(APS sector 13ID)FT-IR microscopy (ALS BL 1.4.3)
- Molecular Dynamic Simulations
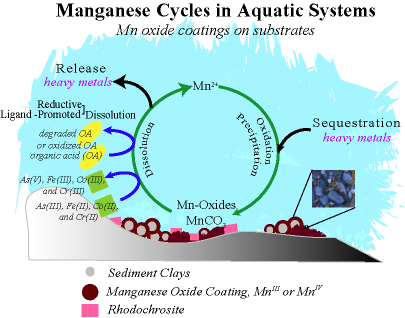
Ph.D. Work: Microscopic Mechanisms of Dissolution and Precipitation of Manganese Minerals
Microscopic Observations of Reductive Manganite Dissolution under Oxic Conditions: Jun, Y. S.; Martin, S. T. Environ. Sci. Technol. 2003, 37, 2363-2370. (Click here to see this manuscript)
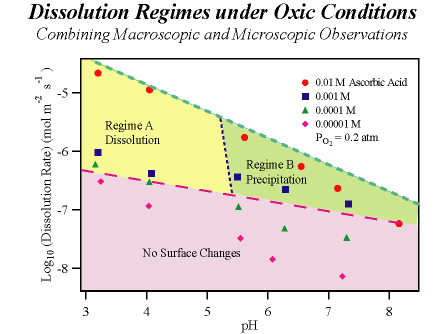
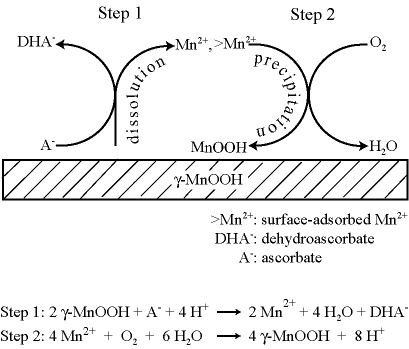
At oxic/anoxic transition zones, manganese release from (hydr)oxide minerals into aqueous solution is a dynamic balance between mineral dissolution and Mn2+(aq) oxidation and precipitation, which are processes respectively promoted by organic reductants and molecular oxygen. We employ a flow-through atomic force microscope reactor (AFM-R) to investigate the reductive dissolution of the {010} surface of manganite (γ-MnOOH) across a range of pH values and ascorbic acid concentrations in aqueous solutions equilibrated with atmospheric CO2 and O2. The apparent dissolution rate increases with higher ascorbic acid concentrations and lower pH values. Concurrent changes in surface morphology show that dissolution proceeds at low pH via etching and step retreat while at high pH dissolution is concurrent with precipitation. The precipitates are characterized ex situ by X-ray photoelectron spectroscopy (XPS) and found to be MnIII-oxide. The onset of precipitation is consistent with an analysis of the thermodynamic driving forces for the reactions of a two step mechanism. In the first step, Mn2+ is released to aqueous solution by reduction of γ-MnOOH in reaction with ascorbic acid. This step is thermodynamically favorable under all conditions employed. In the second step, which leads to precipitation, surface adsorbed Mn2+is oxidized by O2 to yield a MnIII-oxide precipitate. This step is thermodynamically possible only at pH > 5 for our experimental conditions. When the second step is active, the apparent dissolution rate equals the intrinsic dissolution rate minus the precipitation rate. Analysis of the growth rates observed in AFM indicates the precipitation rate reaches 71% of the intrinsic dissolution rate under some reactor conditions. Comparison of our γ-MnOOH results to literature reports for Mn2+ oxidation on γ-FeOOH indicates γ-MnOOH is a more effective surface catalyst by a factor of 108.

AFM deflection-mode micrographs of surfaces changes observed in situ at 298 K during exposure of manganite to ascorbic acid at acidic and neutral pH values. Images {A, B} and {C, D} respectively show surface changes characteristic of regimes A and B in the following figure. (A) pH = 3.2, 10 mM NaNO3. (B) Same as A after exposure to 1 mM ascorbic acid for 95 min. (C) pH = 7.2, 10 mM NaNO3. (D) Same as C after exposure to 10 mM ascorbic acid for 65 min.
Heteroepitaxial Nucleation and Oriented Growth of Manganese Oxide Islands on Carbonate Minerals under Aqueous Conditions: Jun, Y. S.; Kendall, T. A.; Martin, S. T.; Friend, C. M.; Vlassak, J. J. Environ. Sci. Technol. 2005, 39, 1239-1249. (Click here to see this manuscript)
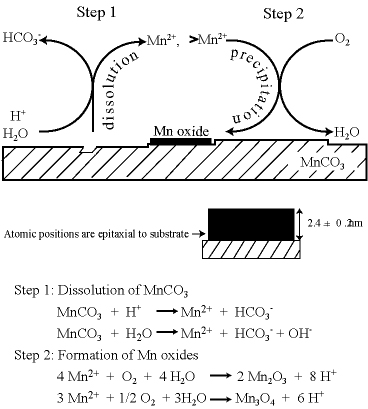
Manganese redox cycling and the accompanying dissolution and precipitation reactions are important processes in natural waters. In the present study, Mn2+(aq) is reacted with O2(aq) at circumneutral pH to form Mn oxide islands on the (10-14) surface of MnCO3. The islands grow heteroepitaxially. The effects of the substrate surface morphology, the substrate atomic structure, and the aqueous concentration of Mn2+ are investigated. On terraces, rhombohedral oxide islands form with 90° rotation relative to the crystallographic axis of the underlying carbonate substrate. Although the island heights self limit between 2 and 3 nm depending on reaction conditions, the islands grow laterally to several square microns before separate islands collide and coalesce. The islands do not grow over substrate steps or down dissolution-pit edges. Comparison studies done with MgCO3 and CaCO3 show that the former also promotes heteroepitaxial growth whereas the latter does not. This difference is explained by the relative bond length mismatch between the structures of the carbonate substrates and the atomic structures of Mn oxides . A free energy model is also presented to explain why the heights of the Mn oxide islands self limit. Our results provide an improved basis both for the development of predictive models of contaminant fate and transport and for the modeling of hydraulic flow through carbonate aquifers.
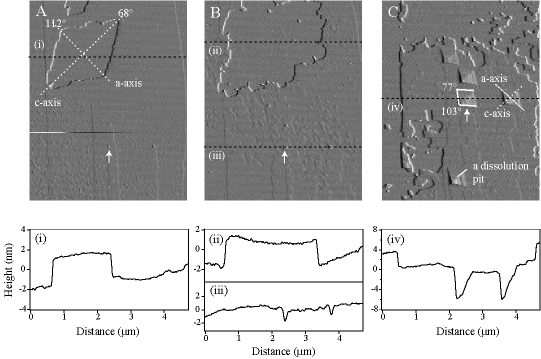
Concurrent island precipitation and substrate dissolution. (A) A rhombohedral island after 120 min exposure to solution. The island grows with 90° rotation relative to the crystallographic axis of the substrate. (B) Growth of the island after 290 min and appearance of a dissolution pit. (C) Coalescence of islands, thus forming a film, after 950 min and contemporaneous appearance of many dissolution pits. The AFM micrographs (4.7 × 6.0 µm2) at 298 K. The solution has pH = 6.1, no added aqueous Mn2+, and 1 atm O2 (aq). The z-scale of the image is 10 nm. The white arrows show a common point of reference in images A to C (image drift).
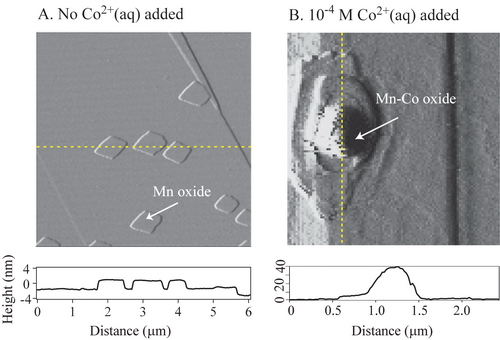
Effects of Cobalt on the Growth of Manganese Oxide Thin Films: Jun, Y. S. and Martin, S. T. Langmuir,2006, 22, 2235-2240 (Click here to see this manuscript)
The mobility of dissolved heavy metals in natural waters is partially regulated by interactions with manganese oxide films. In the current work, the effects of aqueous cobalt(II) on manganese oxide film growth are studied by atomic force microscopy. The film is grown on the (10-14) surface of rhodochrosite ( MnCO3). In the presence of O2(aq) and at circumneutral pH, film growth begins as manganese oxide islands that expand laterally across the surface. Addition of Co2+(aq) leads to the partial or complete dissolution of the manganese oxide film. Simultaneously, there is growth of new islands having multilayer structures that are unrestrained in the z‑direction. The chemical composition of these new islands appears to include both Co and Mn ions. Empirical rules governing the growth of the two types of islands can be developed for the absence and the presence of Co2+(aq). In the absence of Co2+(aq), islands grow as flat two-dimensional rhombohedral islands of nearly uniform height (2.4 ± 0.3 nm). These islands do not cross over steps on the substrate. The growth rules change markedly in the presence of Co2+(aq). The islands grow indefinitely in the z-direction as strata structures of polydisperse thickness and rounded tops. The islands readily grow over steps. Cobalt ions, therefore, relieve the two-dimensional restriction on layer formation and allow three-dimensional growth. Moreover, the shape of the dissolution pits on the surface of MnCO3 changes from rhombohedral in the absence of cobalt to partially rounded in the presence of cobalt. The rounding occurs for the obtuse edges of the pit. Direct microscopic observations of the interactions of cobalt with manganese oxide films provide new mechanistic insights that are important in the quantitative modeling of the mobility of heavy metals in the environment.
Structure and Reactivity of Mn Oxide Film on Carbonate Substrates: Synchrotron-based Measurements:
Y. S. Jun, S. K. Ghose, T. P. Trainor, P. J. Eng, and S. T. Martin, Structure of the Hydrated (10-14) Surface of Rhodochrosite (MnCO3), Environ. Sci. Technol. 2007, 11, 3918-3925. (Click here to see this manuscript)
Manganese oxide films, which form as thin coatings on mineral surfaces, have important consequences in system descriptions of environmental compartments. By insulating the substrate from the aqueous medium, theses films affect alkalinity, the bioavailability of manganese and other divalent cations, and the phase partitioning of toxic heavy metals. Although our results to date have established key information regarding the growth of the Mn oxide film on carbonate minerals, we nevertheless have no direct information about the structure of the film. We have established a hypothesis that the structure of this film should approximate MgCO3 and MnCO3, but not CaCO3 by using atomic force microscopy. We have further hypothesized that the atomic positions in this nanophase film differ from their bulk counterparts by observing the self limiting height growth. We have been measuring synchrotron-based crystal truncation rods (CTRs) and glazing incidence X-ray absorption fine structure (GI-XAFS) spectroscopy to determine the surface structure of the substrate and the Mn oxide film. Moreover, given our extensive knowledge of this films properties based upon atomic force microscopy observations and the effects of saturation state on film growth, the structural knowledge gained from the synchrotron-based measurements will be highly leveraged towards the development of a much more comprehensive understanding of the film growth and reactivity.

